RESEARCH

The subependymal zone (SEZ) is the largest germinal region in the adult rodent brain producing millions of neurons that migrate towards the olfactory bulb during the lifespan. In this neurogenic region, neural stem cells (NSCs) reside in the walls of the lateral ventricles in a highly specialized microenvironment known as ‘neurogenic niche’. This finely tuned cellular ecosystem displays a specific cytoarchitecture and a complex network of molecular and cellular components that actively participate in NSC maintenance and regulation.
Our main focus of research is the study of the intrinsic and extrinsic signals that influence NSC behavior, paying special attention to the understanding of different cues that control the dynamic transitions among quiescent, primed-for-activation and activated NSC states. Additionally, we work to elucidate the regulation of the symmetric/asymmetric division of NSCs as one of the crucial mechanisms that ensures their self-renewal while maintaining the constant generation of differentiated progeny.
We could subdivide our research focus into the following specialized areas:
Intrinsic regulation of NSCs
We are interested in understanding the cell-autonomous regulators that control stemness and neurogenic potential. In this context, in the last decades, we have described diverse intrinsic molecules that play an important role in these processes such as the kinase Dyrk1a, the cell cycle inhibitors and transcriptional regulators p21 and p27 or the membrane metalloproteinase MT5-MMP that cleaves the N-cadherin anchor of NSCs to the niche.

Extrinsic signaling by the SEZ niche
Different cellular components in the SEZ co-exist with NSCs, being essential for their maintenance and regulation. Among the different elements in the niche, we have specially focused on the regulation mediated by vasculature, microglia and neuronal innervation. Indeed, NSCs have a unique disposition and interaction with other cell constituents, specially vasculature. NSCs are localized around blood vessels (BV), which renders them susceptible both to soluble (locally secreted and blood-borne) and to adhesion-related (cell-cell and cell-ECM) cues. Our laboratory has identified the first angiocrine factor described to be involved in NSC regulation, the epithelium-derived growth factor (PEDF). Furthermore, we also reported the role of vascular secreted neurotrophin-3 (NT-3) in the maintenance of NSC quiescence. Regarding innervation, we reported an essential role for the protein α-synuclein, present in dopaminergic nigral afferents, in the regulation of adult NSCs, identifying the first synaptic regulator with an implication in the stem cell niche biology.

Neuroimmune interactions
There is growing evidence in the field that the immune system participates in the maintenance of the brain homeostasis and function, and that also has a role in regulating adult neurogenesis. This has expanded the concept of niche, evidencing its integration in the homeostasis of the whole organism. In this way, we have recently identified the role of peripherally-induced inflammation in regulating NSC activation state though TNF-α receptors 1 and 2 and we are currently exploring other interactions between NSCs and the immune system.
Aging and neurodegeneration
Apart from the study of NSC in its native niche, we are interested in understanding the changes that occur in an aging brain to elucidate how this affects NSCs and its neurogenic potential. Specifically, we study senescence as a non-pathological aging process that strongly affects neurogenesis. As for pathological scenarios, we are also studying how proteins involved in neurodegenerative diseases, such as the accumulation of misfolded α-synuclein observed in Parkinson Disease, may affect the normal NSC functioning.

After years of collaboration and several independent projects on previous GVA PROMETEO calls, the Molecular Neurobiology Lab (PI: Isabel Fariñas) and the Cell Cycle Lab (PI: Juan Carlos Igual), both from the University of Valencia, have joined forces and obtained a grant in the 2020 ‘Programa PROMETEO para grupos de investigación de Excelencia’ from Generalitat Valenciana.
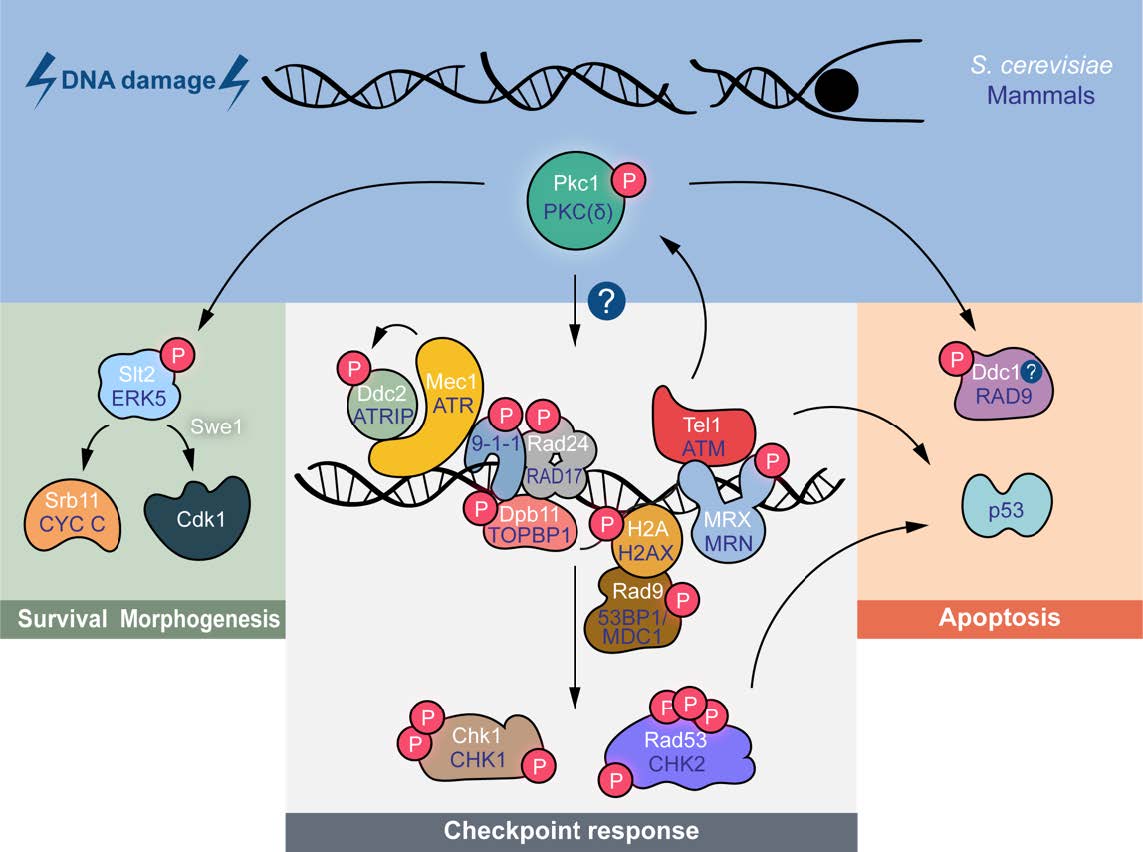
Together we are going to explore the mechanisms that control de DNA damage checkpoint, and specially the involvement of PKC proteins, using a multidisciplinary approach in both murine Neural Stem Cells (NSCs) and Saccharomyces cerevisiae. Additionally, the project will study other aspects related to the genomic stability of NSCs such as their malignant transformation (as glioma initiating cells) and their mechanisms to evade the immune system surveillance.
Project Title: Una aproximación multidisciplinar al estudio de la respuesta al daño genómico en células madre neurales: de levaduras a mamíferos y vuelta.

A MULTIDISCIPLINARY APPROACH TO EXPLORING DNA DAMAGE RESPONSES IN QUIESCENCE: FROM YEAST TO MAMMALIAN NEURAL STEM CELLS AND BACK
The NeuroYeast consortium was consolidated through a successful collaboration within the Prometeo program, which united complementary expertise to identify conserved regulators of the DNA damage response, including protein kinase C isoforms such as PKCδ. This joint effort established a powerful dual platform based on iterative experimentation between yeast and mammalian stem cells, enabling comprehensive mechanistic analyses across model systems.
Building on this foundation, we now investigate how quiescent cells respond to genotoxic stress, addressing reversible quiescence as a key biological state linked to stem cell maintenance, exhaustion, and cancer. In particular, we study neural stem cells and their malignant counterparts, glioma stem cells, which often resist therapy through quiescence associated mechanisms. By combining large scale unbiased molecular approaches with candidate driven research, we aim to uncover conserved mechanisms governing the DNA damage response in the context of cellular quiescence, with broad biomedical relevance.